Drug Impurity Reference Standards For Sale
Drug Impurity Reference Standards for Accurate Pharmaceutical Analysis
– Ensure regulatory compliance and analytical accuracy with Hipsoul’s certified drug impurity reference standards.
– Each compound is fully characterized by LC-MS, NMR, and HPLC, and supplied with COA, SDS, and full traceability.
– Available in stock for global delivery.

Some Questions You Should Concern Before Order
Are these impurity standards ICH Q3A/B and USP compliant?
Yes. All Hipsoul impurity reference standards are developed and characterized in accordance with ICH Q3A/B, USP, and EP guidelines for impurity qualification.
Each compound is fully documented with regulatory references, ensuring compliance for use in pharmaceutical R&D, QC, and method validation.
Do you provide COA, NMR, LC-MS, and HPLC data for each compound?
Absolutely. Every impurity standard is supplied with a comprehensive Certificate of Analysis (COA), including:
- Structural confirmation by NMR (¹H & ¹³C)
- Mass verification by LC-MS or HRMS
- HPLC purity profile (≥ 99%)
- Storage and stability recommendations
This guarantees traceability, reliability, and full analytical transparency for your documentation and audits.
Do you provide COA, NMR, LC-MS, and HPLC data for each compound?
Yes. Hipsoul provides custom synthesis services for new or rare impurities, degradation products, and metabolites.
Our experienced chemists can design, synthesize, and characterize compounds upon request — delivering analytical-grade materials with complete COA, NMR, and LC-MS data.
Typical turnaround time: 3–6 weeks, depending on structure complexity.
How long does delivery take for in-stock and custom standards?
- In-stock impurity standards: Usually ship within 3–5 business days after order confirmation.
- Custom-synthesized standards: Typically 3–6 weeks, including synthesis, purification, and full analytical verification.
Global shipping is available with temperature-controlled packaging to ensure stability during transit.
What’s the purity and stability of your impurity materials?
All Hipsoul impurity standards are analytically verified at ≥ 99% purity by HPLC and LC-MS.
Stability is evaluated under controlled storage conditions, and recommended temperature and shelf-life are clearly stated on the COA.
Each batch undergoes retesting and revalidation to maintain long-term accuracy and consistency for regulated use.
Showing 1–8 of 15 results
-
Drug Impurity Reference Standards
Adenosine 5’-triphosphate Disodium Salt Reference Standard Material for Sale
Rated 0 out of 5$89.00Original price was: $89.00.$69.00Current price is: $69.00. -
Best Selling Products
Albendazole Reference Standard Material for Sale
Rated 0 out of 5$89.00Original price was: $89.00.$69.00Current price is: $69.00. -
Drug Impurity Reference Standards
Butafosfan Reference Standard Material for Sale
Rated 0 out of 5$89.00Original price was: $89.00.$69.00Current price is: $69.00. -
Drug Impurity Reference Standards
Calcium Gluconate Reference Standard Material for Sale
Rated 0 out of 5$89.00Original price was: $89.00.$69.00Current price is: $69.00. -
Drug Impurity Reference Standards
Clemastine Fumarate Reference Standard Material for Sale
Rated 0 out of 5$89.00Original price was: $89.00.$69.00Current price is: $69.00. -
Drug Impurity Reference Standards
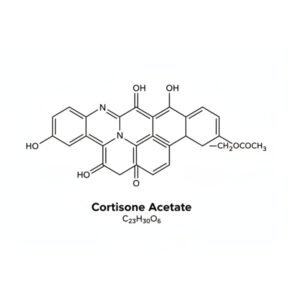
Cortisone Acetate Reference Standard Material for Sale
Rated 0 out of 5$89.00Original price was: $89.00.$69.00Current price is: $69.00. -
Drug Impurity Reference Standards
Doxycycline Hydrochloride Reference Standard Material for Sale
Rated 0 out of 5$89.00Original price was: $89.00.$69.00Current price is: $69.00. -
Drug Impurity Reference Standards
Florfenicol Reference Standard High-Purity Reference Materiel for Sale
Rated 0 out of 5$89.00Original price was: $89.00.$69.00Current price is: $69.00.
Some Features of Drug Impurity Reference Standards
Hipsoul’s Drug Impurity Reference Standards are designed to help pharmaceutical laboratories achieve analytical accuracy, regulatory compliance, and traceable results across LC-MS, GC-MS, and HPLC systems.
🔬 Key Features
- Certified Purity ≥ 99% – Each impurity is fully characterized by LC-MS, NMR, and HPLC to ensure analytical reliability.
- Regulatory Compliance – Developed in alignment with ICH Q3A/B, USP, and EP impurity guidelines.
- Comprehensive Documentation – Supplied with COA, SDS, and analytical spectra for every batch.
- Traceable & Reproducible – Batch-to-batch consistency with full traceability records for audits and submissions.
- Custom Synthesis Service – Fast development of novel impurities, metabolites, or degradation products not commercially available.
- Stability-Assured Packaging – Sealed vials with clear labeling, recommended storage conditions, and expiration dates.
- Global Availability – In-stock compounds ready for worldwide shipment with temperature-controlled logistics.
- Expert Technical Support – Direct assistance from experienced analytical chemists for method validation and troubleshooting.
Types of Drug Impurity Reference Standards We Supply
- Albendazol
- Florfenicol
- Levamisole Hydrochloride
- Praziquantel
- Moxidectin
- Sodium citrate
- Cortisone acetate
- Doxycycline Hydrochloride
- Clemastine Fumarate
- Procaine hydrochloride
- Magnesium L-aspartate
- Calcium gluconate
- Butafosfan
- Adenosine 5’-triphosphate disodium salt
- Uridine 5'-triphosphate trisodium salt(UTP-Na3)
- Glutathione(G-SH)
Types of Drug Impurity Reference Standards We Supply
Choosing the right supplier for impurity reference materials is critical to analytical reliability, regulatory compliance, and long-term partnership success.
Here’s why global pharmaceutical and analytical labs trust Hipsoul:
🔬 1. Scientifically Verified Purity and Identity
Every impurity standard is rigorously characterized by LC-MS, NMR, HPLC, and other orthogonal techniques.
Each compound is analytically confirmed for purity ≥ 99 %, with full data traceable to its batch record.
🧾 2. Complete Regulatory Documentation
All materials are supplied with a comprehensive Certificate of Analysis (COA), SDS, and spectral data—meeting ICH Q3A/B, USP, and EP guidelines for impurity qualification and validation studies.
⚗️ 3. Custom Synthesis & Rare Impurity Solutions
If a target impurity is not available in stock, Hipsoul’s chemists can design and synthesize custom impurities, metabolites, and degradation products within 3–6 weeks, delivering analytical-grade compounds with verified structure and purity.
🚀 4. Fast Delivery & Global Support
In-stock impurity standards ship within 3–5 business days, with temperature-controlled logistics and export documentation.
Our technical team provides 24-hour response for quotations, COA requests, and method-development support.
🧠 5. Expert Team with Analytical Insight
Hipsoul’s R&D and QA teams combine expertise in analytical chemistry, organic synthesis, and pharmaceutical regulation, ensuring your laboratory receives not just materials—but scientific support for every analytical challenge.
🌍 6. Trusted by Global Laboratories
Pharmaceutical companies, CROs, and QC labs worldwide rely on Hipsoul standards for method validation, impurity profiling, and stability testing—backed by consistent quality and dependable documentation.
Hipsoul delivers certified, traceable, and fully characterized impurity standards — empowering your lab to achieve accuracy, compliance, and confidence in every analytical result.