Cortisone Acetate Reference Standard Material for Sale
Original price was: $89.00.$69.00Current price is: $69.00.
+ Free Shipping- Product Code: C-D25007X
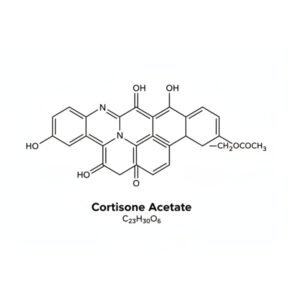
- Chemical Name: Cortisone Acetate
- Category: Drug Impurity Standard
- CAS Number: 50-04-4
- Molecular Formula: C₂₃H₃₀O₆
- Molecular Weight: 402.48 g/mol
- Purity: ≥ 95%
- Available Package Size: 25 mg
- Storage Conditions: 2–8 ℃
- Special Requirements: None
1️⃣ Product Description
Product Name: Cortisone Acetate Reference Standard
CAS Number: 50-04-4
Product Code: C-D25007X
Category: Drug Impurity Standard
Short Overview:
Cortisone Acetate (CAS 50-04-4) is a high-purity drug impurity reference standard (≥ 95 %) used for analytical testing and pharmaceutical quality control.
It is widely employed in HPLC, LC-MS, and GC analysis for impurity profiling, method validation, and stability studies of corticosteroid formulations.

Information:
| Parameter | Value |
|---|---|
| Molecular Formula | C₂₃H₃₀O₆ |
| Molecular Weight | 402.48 g/mol |
| Purity | ≥ 95 % |
| Storage Conditions | 2 – 8 °C |
| Package Size | 25 mg |
| Documentation | COA & SDS supplied |
| Compliance | ISO/IEC 17025 & GMP procedures |
💡 Stable under refrigerated conditions and classified as non-hazardous for international shipping.
2️⃣ Features & Benefits / Technical Information
Features & Benefits
- ✅ Analytical-grade purity ≥ 95 %, verified by LC-MS and HPLC
- ✅ Supplied with Certificate of Analysis (COA) and Safety Data Sheet (SDS)
- ✅ Fully traceable batch documentation for regulatory compliance
- ✅ Manufactured under ISO/IEC 17025 and GMP quality systems
- ✅ Stable at 2–8 °C for long-term reliability
- ✅ Fast global delivery and custom sizes available
Technical Information
| Parameter | Details |
|---|---|
| Structural Formula | (Available upon request or shown as image) |
| Appearance | White to off-white crystalline powder |
| Storage Conditions | 2–8 °C (Refrigerated, Dry Environment) |
| Shelf Life | 24 months from manufacture date |
| Analytical Techniques | HPLC, LC-MS, GC |
| Category | Drug Impurity Standard |
📎 Each batch is tested for identity and purity by HPLC and LC-MS before release.
3️⃣ Applications
Cortisone Acetate reference standard is commonly used in:
- Pharmaceutical Quality Control: Detection and quantification of impurities in corticosteroid drugs.
- Stability Studies: Identification of degradation products during shelf-life testing.
- Analytical Method Validation: Calibration and verification of HPLC and LC-MS systems.
- Research & Development: Drug metabolism and pharmacokinetic studies.
💡 Ensures precision, traceability, and regulatory compliance for steroid impurity analysis.
4️⃣ Frequently Asked Questions (FAQs)
Q1. What analytical methods are compatible with Cortisone Acetate standards?
HPLC, LC-MS, and GC methods are recommended for qualitative and quantitative impurity testing.
Q2. Do I receive COA and SDS with my order?
Yes, each shipment includes a Certificate of Analysis and Safety Data Sheet.
Q3. Can I request custom sizes or bulk quantities?
Yes, we offer custom packaging and bulk orders on request.
Q4. How should I store this product?
Store tightly sealed at 2–8 °C in a dry environment away from light and humidity.
Q5. Is this product hazardous for shipping?
No, Cortisone Acetate reference standard is classified as non-hazardous for air and ground transport.
5️⃣ Ordering & Support
| Product Code | CAS | Size | Availability |
|---|---|---|---|
| C-D25007X | 50-04-4 | 25 mg | ✅ In Stock |
📦 Delivery: Ships within 3–5 business days worldwide.
🧾 Includes: COA & SDS with each shipment.
🌍 Global Shipping: Temperature-controlled logistics available for sensitive materials.
📩 Contact Sales: sales@hipsoulab.com
📞 Customer Support: +86-755-28502380
💬 Hipsoul — Delivering purity, precision, and reliability to analytical laboratories worldwide.
You must be logged in to post a review.



Reviews
There are no reviews yet.